To begin, complete the form below.
Transcranial Magnetic Stimulation (TMS) therapy, is an FDA-approved, non-invasive treatment that uses targeted magnetic pulses to stimulate specific areas of the brain involved in mood regulation.
It does not require anesthesia. It does not involve systemic medication. Patients remain awake and alert throughout treatment.
TMS is most often considered for major depressive disorder when adequate trials of antidepressant medication have not led to meaningful improvement. In some cases, it may also be considered for obsessive-compulsive disorder.
This is an advanced treatment option. It is used thoughtfully and selectively.
It is not typically used as a first-line treatment.
TMS is also not appropriate for everyone. Certain medical conditions and implanted devices may limit eligibility. A structured evaluation is required before proceeding.
Our role is to determine whether TMS fits your diagnosis, history, and goals. Not simply whether it is available.
TMS delivers focused magnetic pulses through a specialized treatment device positioned over the scalp.
These pulses create small electrical currents in targeted brain regions associated with mood and behavioral regulation. Over a series of treatments, this stimulation can help strengthen neural pathways that may be underactive in depression.
The treatment is localized. It does not circulate through the body in the way medication does.
Changes occur gradually over several weeks.
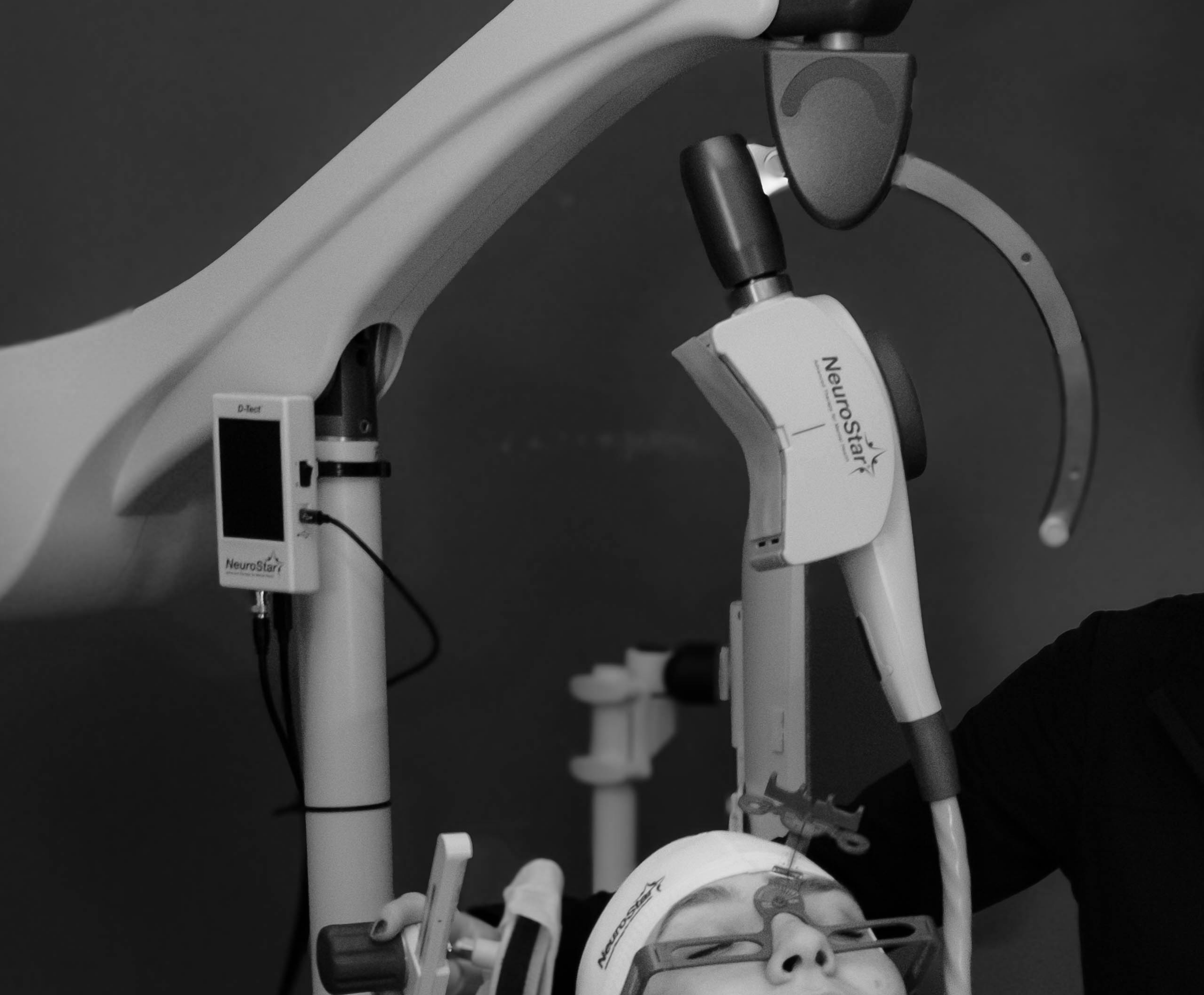
Treatment typically occurs five days per week during the acute phase, often over four to six weeks. Individual schedules vary based on protocol and clinical response.
Each session is brief, usually around 20 minutes. Patients remain awake and can return to normal daily activities afterward.
Progress is monitored throughout the course of treatment. We track symptom changes carefully and make adjustments when clinically indicated.
We will provide a detailed TMS “What to Expect” guide prior to starting treatment.
TMS is generally well tolerated. The most common side effects include:
Mild scalp discomfort
Headache
Temporary sensitivity at the treatment site
Not all patients respond. Some require maintenance planning. Others may transition to different strategies if response is limited.
Careful screening reduces risk and increases the likelihood of benefit.
Degree of symptom improvement
Ongoing medication needs
Maintenance or relapse prevention strategies
For some patients, TMS reduces the need for additional medication adjustments. For others, it creates momentum that supports other therapies more effectively.
The goal is sustained progress, not temporary response.
Many insurance plans cover TMS for treatment-resistant depression when criteria are met. We review eligibility before starting.
Unlike electroconvulsive therapy, TMS does not cause memory loss.
Most patients describe a tapping sensation on the scalp during stimulation. Discomfort, when present, is typically mild and improves over time.
We coordinate with specialists and other healthcare providers to ensure comprehensive care for all aspects of your health.
Visit our Resources page for additional patient education documents and TMS-specific resources.
If depression has not improved despite appropriate treatment, we can evaluate whether TMS is a reasonable next step.
Advanced treatments should be introduced carefully, with full understanding of risks, limits, and expectations.